 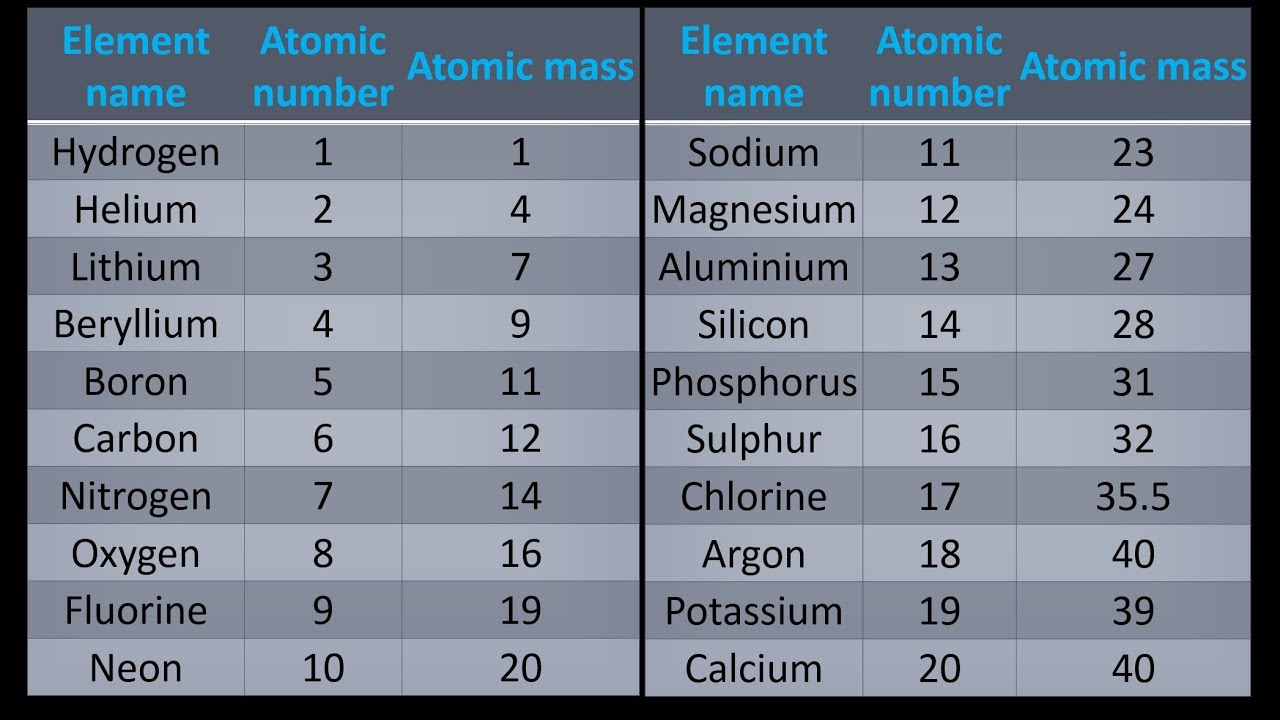
(Yes, the number 12.01 has the right number of significantfigures, even though 1.11% only has 3 significant figures.) The average mass is simply a weighted average. The mass spectrometer reports that there are two isotopes ofcarbon, 98.99% of the sample has a mass of 12 amu ( not a surprise since this is the atom on which the scale is based).ġ.11% of the sample has a mass of 13.003355 amu ( this isotope is 1.0836129 times as massive as 12C) The average mass of a carbon iscalculated from the information the mass spectrometer collects. 14C dating is accomplished by measuring the radioactivity of a sample, not by actually counting the number of 14C atoms. The natural abundances of 14C, 10C and 11C are so low that most mass spectrometers cannot detect the effect these isotopes have on the average mass. If a sample of carbon was placed in a mass spectrometer thespectrometer would detect two different C atoms, 12C and 13C. No single carbon atom has a mass of 12.01 amu, but in a handful of C atoms the average mass of the carbon atoms is 12.01 amu. This is the average atomic mass of carbon. 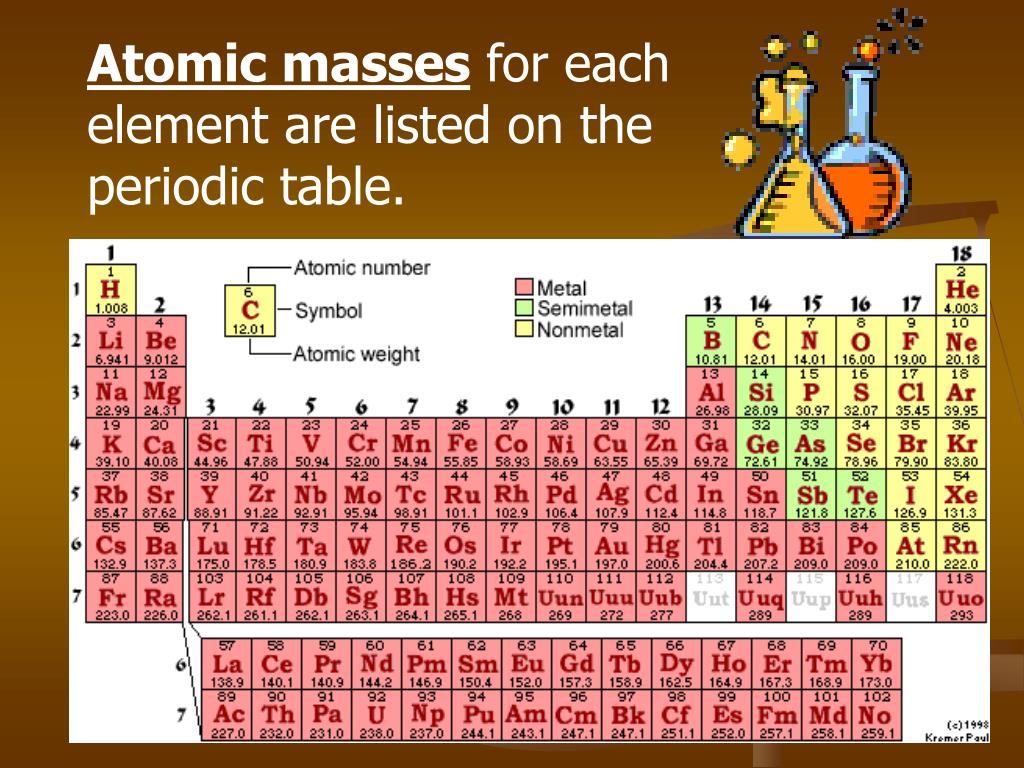
On the periodic table the mass of carbon is reported as 12.01 amu. Since many elements have a number of isotopes, and since chemistsrarely work with one atom at a time, chemists use average atomicmass. Since there are a variety of carbon isotopes we must specify which C atom defines the scale.Īll the masses of the elements are determined relative to 12C.īy the way, the mass of an element is not equal tothe sum of the masses of the subatomic particles of which the elementis made! 12C has 6 neutrons, 13C has 7 neutrons, and 14C has 8 neutrons and so on. Each carbon atom has the same number of protons and electrons, 6. Carbon exists as two major isotopes, 12C, and 13C ( 14C exists and has a half life of 5730 y, 10C and 11C also exist their half lives are 19.45 min and 20.3 days respectively). Why do we specify 12C? We do not simply state the themass of a C atom is 12 amu because elements exist as a variety ofisotopes. 
Multiply the number of each atom by its atomic weight (found on the Periodic Table of the Elements).Atomic mass is based on a relative scale and the mass of 12C (carbon twelve) is defined as 12 amu. Water is made up of 2 hydrogens and 1 oxygen.Ģ. The molar mass is calculated by taking the sum of the atomic weights of all the atoms which form the molecule.įirst determine the number of each atom in the formula.ġ. When there is parenthesis's around a group of atoms we must multiply each atom by the subscript to get the total number of each element.Ĭa(NO 3) 2 1 Calcium 2 Nitrogen and 6 Oxygen -> 3 x 2 Reading Compounds that have (parenthesis's). If there is only 1 atom we do not use subscripts. The subscript belongs to the element it follows and indicates how many of that atom there are. 
Reading Simple Chemical Formulas- Each element begins with a CAPITAL letter. The units are atomic mass units ( amu ).ġamu =1.660 538 782×10 –27 kg 1 amu is 1/12 the mass of a carbon-12 atom which has a mass of 12.0000. Molecular Mass is the mass of a given molecule (NOT MOLES of molecules). Molecular Weight is the molar mass of a COVALENT compound. The unit for molar mass (note it is the mass of a mole) is grams/mole.Ītomic Weight is the molar mass of an element.įormula Weight is the molar mass of an IONIC compound. Molar Mass is the mass of one mole of a substance (6.02 x 10 23 formula units). (Molar Mass, Molecular Weight, Gram Formula Mass) 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
